
Two of the possible aldotetroses are D sugars, while the other two are L sugars. Aldotetroses have two chiral centers and hence possess 2 2 = 4 stereoisomers, or two pairs of enantiomers. Recall that a given organic molecule has 2 n stereoisomers, where n is the number of chiral centers. By the same token, a L sugar does not always rotate polarized light counterclockwise all the L prefix does is indicate that the molecule is the enantiomer of the corresponding R sugar. In view of these exceptions, the D notation in the context of monosaccharides no longer connotes the direction to which the molecule displaces polarized light rather, it becomes simply an indicator of the fact that the lowermost chiral center has the R configuration, just as it does in (+)-glyceraldehyde. Examples include D-erythrose, which has specific rotation = –32.7 o (recall that the negative sign denotes counterclockwise rotation), and D-threose, for which = –12.2 o. Importantly, while D-glyceraldehyde is dextrorotatory, the D enantiomers of some sugars do not rotate plane-polarized light in a clockwise sense. 
Note how in all three molecules the lowermost chiral center has a hydroxyl group pointing to the right. Shown below are the Fischer projections of D-glucose, D-fructose, and D-ribose, a common aldopentose. In accord with this convention, almost all naturally occurring carbohydrates are D-sugars, which means that the chiral center farthest from the carbonyl group will have an OH group pointing to the right in the Fischer projection. In response to this finding, chemists began using the Fischer-Rosanoff convention whereby the prefix D– is attributed to any sugar that degrades to (+)-glyceraldehyde. On the other hand, when synthetic sugars are degraded, they produce a mixture of D– and L-glyceraldehyde. 
Most naturally occurring monosaccharides can undergo a similar degradation process. One example is glucose, which can be stripped of three carbons, from the top to the bottom of the Fischer projection, to yield D-glyceraldehyde. It can be shown that most naturally occurring carbohydrates can be degraded (broken down) to produce D-glyceraldehyde. The D enantiomer is the only form abundant in nature. The (+) or D– designations indicate that the enantiomer rotates plane-polarized light in a clockwise fashion (dextrorotatory), while the (-) or L designations are applied to the enantiomer that rotates plane-polarized light in a counterclockwise (levorotatory) sense. One of the smallest compounds considered to be a carbohydrate is glyceraldehyde, which has a chiral center and therefore can exist as a pair of enantiomers, as shown. Glucose is an aldohexose, while fructose is a ketohexose the suffix – ose indicates that the molecule is a sugar. Glucose is an example of aldose, while fructose is an example of ketose.Īldoses and pentoses can be further classified on the basis of their number of carbons, using the typical prefixes tetr- (four carbons), pent– (five carbons), hex- (six carbons), and so forth. Simple monosaccharides can be classified as aldoses, when they contain an aldehyde group, or ketoses, when they contain a ketone group. 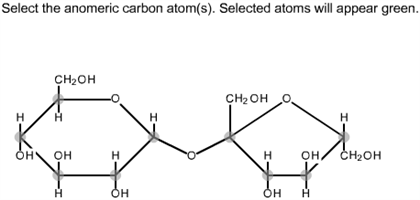
In this article, I provide a brief introduction to the classification and representation of simple aldoses, carbohydrates with an aldehyde group and no more than five or six carbons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
